Why Galvanic Corrosion Occurs on Silver-Plated PCBs and How to Prevent It
Silver plating, a common technique in PCB manufacturing, is widely used to enhance solderability and improve the contact performance of copper pads. This thin layer of silver provides a smooth and reliable surface for component mounting, ensuring a strong electrical connection. However, beneath the surface benefits lies a hidden challenge: the Galvani Effect. When copper and silver meet, a galvanic reaction can occur, especially in the presence of moisture, leading to accelerated corrosion. This subtle yet impactful phenomenon can compromise the integrity of the PCB and its long-term reliability. Understanding how this reaction unfolds is key to preventing damage and optimizing the performance of silver-plated PCBs.
What is the Galvani Effect?
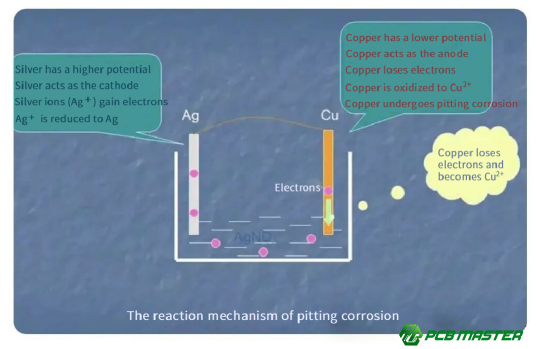
The Galvani Effect, also known as galvanic corrosion, occurs when two different metals come into contact in the presence of moisture or an electrolyte. This causes one metal to corrode faster than it would if it were alone. The metals form a small electrical cell, known as a galvanic cell, where one metal acts as the anode and corrodes, while the other metal serves as the cathode and is protected.
Electrochemical Corrosion and Electrical Potential Difference
Galvanic corrosion is based on the principle of electrochemical corrosion. Metals, when exposed to different environmental conditions, develop an "electrical potential" — a measure of their ability to lose electrons. If two metals with different electrical potentials are connected, electrons will flow from the metal with the lower potential (anode) to the metal with the higher potential (cathode). This electron flow causes the anode to undergo corrosion.
For example, in a copper-silver system, copper has a lower electrical potential than silver. When these two metals are in contact, the copper corrodes because it loses electrons, while the silver remains largely unaffected.
Origin and Naming: Luigi Galvani's Discovery
The term "Galvani Effect" is named after the Italian scientist Luigi Galvani, who first discovered the phenomenon in the 18th century. Galvani was studying the effects of electrical currents on animal tissue when he observed that a frog's leg twitched when touched by two different metals. He realized that the metals were generating an electric current, which led to the concept of bioelectricity. This principle was later applied to metal corrosion in engineering and materials science, giving rise to the term "galvanic corrosion."
Today, the Galvani Effect is a critical concept in materials science, particularly in understanding how different metals interact in various environments, such as in printed circuit boards (PCBs), where copper and silver are often used together.

What is the Principle Behind Galvani Effect in Silver-Plated PCBs?
The Galvani Effect in silver-plated PCBs occurs when the copper (anode) and silver (cathode) form a galvanic cell, causing the copper to corrode faster in the presence of moisture or electrolytes due to their different electrical potentials.
Composition of Silver-Plated PCBs
Silver-plated PCBs are made by applying a thin layer of silver onto the copper pads of the PCB. The structure of these boards consists of a copper base, which is a good conductor of electricity, and a thin silver layer, which enhances solderability and electrical contact performance. The copper provides mechanical strength and electrical pathways, while the silver layer helps with better connection during component mounting.
The relationship between copper and silver in this structure is important because, when exposed to certain environmental conditions, these two metals create a galvanic reaction. Copper, with a lower electrical potential, acts as the anode, while silver, with a higher electrical potential, acts as the cathode.
Silver Plating Process and Purpose
The silver plating process involves applying a thin silver coating onto copper using chemical deposition techniques. This plating improves the PCB’s overall performance by making it easier to solder components and ensuring a stable electrical connection. The primary goal is to protect copper from oxidation, increase the board’s durability, and improve its resistance to corrosion. However, despite these benefits, the silver layer also introduces a risk of galvanic corrosion when exposed to moisture or contaminants.
Conditions for the Galvani Effect to Occur
For the Galvani Effect to occur, certain conditions must be met:
l Electrochemical Potential Difference Between Materials: Silver and copper have different electrical potentials. Silver has a higher potential (+0.80V) than copper (+0.34V). When these metals are in contact, electrons flow from the copper (anode) to the silver (cathode), causing the copper to corrode.
l Role of Electrolytes: Moisture and contaminants in the environment act as electrolytes that trigger the galvanic corrosion. Airborne contaminants like chloride and sulfur ions (from humidity or human contact) can form a thin electrolyte layer on the PCB’s surface, setting the stage for the corrosion process.
Triggers of the Corrosion Reaction
Corrosion occurs when small imperfections or gaps in the silver layer allow moisture and electrolytes to reach the copper underneath. These gaps, which can form at the edges of the silver layer or in tiny holes, create areas where the copper can directly interact with the electrolyte, forming a tiny galvanic cell.
l Microscopic Pores and Gaps: The silver layer, though thin, is often not completely flawless. Small pores or gaps in the silver plating allow the electrolyte to penetrate and reach the copper, where the corrosion process begins.
l Formation of a Galvanic Cell: When the copper and silver are in contact, the presence of moisture or electrolytes enables the formation of a small galvanic cell. The copper at the anode undergoes oxidation, releasing copper ions into the electrolyte, while the silver at the cathode is protected. The corrosion of copper accelerates as the galvanic reaction continues, leading to damage over time.
This process is the core reason why silver-plated copper pads on PCBs are vulnerable to accelerated corrosion when exposed to moisture or environmental contaminants.
What is the Corrosion Mechanism of the Galvani Effect?
The corrosion mechanism of the Galvani Effect involves copper (the anode) undergoing oxidation and dissolving into the electrolyte, while silver (the cathode) remains protected, leading to the buildup of corrosion products that can damage the PCB structure.
Corrosion Process of Copper (Anode)
The corrosion of copper occurs when it acts as the anode in a galvanic cell, where it loses electrons and undergoes an oxidation reaction. In this process, copper atoms on the surface give up electrons and become copper ions (Cu²⁺). These electrons flow through the metal, moving towards the silver layer (the cathode). The copper ions then dissolve into the surrounding moisture or electrolyte. This dissolution of copper accelerates corrosion because the material is being lost, weakening the PCB structure.
For example, when copper is exposed to an electrolyte, such as moisture containing chloride ions, it oxidizes:
Cu → Cu²⁺ + 2e⁻
The copper ions (Cu²⁺) are then carried away by the electrolyte, which contributes to the material's degradation over time.
Protective Role of Silver (Cathode)
Silver, as the cathode, does not corrode but instead participates in a reduction reaction. It attracts the electrons from the copper anode and uses them for a process called oxygen reduction. In the presence of oxygen and water, this reduction reaction forms hydroxide ions (OH⁻) on the surface of the silver. These hydroxide ions help to neutralize the corrosion process by reducing the copper ions in the electrolyte.
The typical oxygen reduction reaction on the silver surface is as follows:
O₂ + 2H₂O + 4e⁻ → 4OH⁻
By consuming the electrons, silver remains protected, but the overall process accelerates the corrosion of copper as it is the one losing material.
Formation and Impact of Corrosion Products
As copper ions (Cu²⁺) dissolve into the electrolyte, they eventually react with hydroxide ions (OH⁻) generated at the silver cathode. This reaction forms copper hydroxide (Cu(OH)₂), which can then further oxidize to copper oxide (CuO). These copper corrosion products, particularly copper salts, have a larger volume than the original copper metal.
The expansion of these corrosion products can cause serious damage:
l Copper Salt Expansion: Copper salts have a much larger volume than the original copper, and their buildup beneath the silver layer creates pressure. This pressure can cause the silver layer to lift or peel off from the copper surface.
l Damage to Silver Layer: The mechanical stress from the expanding corrosion products leads to the silver layer becoming detached or cracked, exposing the copper underneath to further corrosion. This cycle accelerates and leads to the progressive failure of the PCB.
This phenomenon is a critical issue in maintaining the structural integrity of silver-plated PCBs, as it weakens the connection between layers and damages the functionality of the electronic board.
What Is the Impact of the Galvani Effect on PCB Reliability?
The Galvani Effect negatively impacts PCB reliability by causing silver layer blistering and peeling, leading to electrical failures, increased contact resistance, and potential open circuits that can result in system malfunction or complete failure.
Silver Layer Blistering and Peeling
The Galvani Effect can lead to the silver layer on a PCB blistering and peeling off. This occurs because the corrosion products formed underneath the silver layer expand and exert pressure on the silver surface. As copper ions dissolve and combine with hydroxide ions to form copper salts, these corrosion products increase in volume. The buildup of pressure from these expanding products can cause the silver layer to detach or lift from the copper pad.
Once the silver layer is compromised, it loses its ability to protect the underlying copper, accelerating the corrosion process further. The physical damage to the silver layer not only affects the board’s structural integrity but also compromises its electrical performance, making the board more prone to failure.
Damage to Electrical Performance and Integrity
When the silver layer lifts or peels away, the exposed copper pad becomes vulnerable to further corrosion and degradation. This breach in the protective layer leads to an increase in electrical resistance, which can cause poor electrical contact. This degradation directly affects the PCB's ability to conduct signals or power efficiently, making it more susceptible to malfunction or complete failure in an electronic circuit.
Electrical Failures and Circuit Damage
The Galvani Effect can cause significant electrical failures and damage to the PCB’s circuitry. As the copper corrodes under the silver layer, it weakens the PCB's ability to provide reliable electrical connections between components.
l Pad Damage and Increased Contact Resistance: The corrosion of copper beneath the silver layer often leads to damage in the copper pads. This can result in higher contact resistance, which negatively impacts the PCB's ability to transmit electrical signals or power efficiently. Over time, this resistance can lead to overheating or other operational issues.
l Formation of Open Circuits and System Failure: In severe cases, the corrosion can lead to open circuits, where the electrical connection is completely broken. This can occur when the copper beneath the silver layer deteriorates to the point where it no longer forms a functional path for current to flow. Once an open circuit forms, the PCB is effectively damaged and may no longer function as intended, causing system failures or breakdowns in the overall electronic system.
The Galvani Effect significantly reduces the lifespan and reliability of PCBs, particularly in environments where moisture or contaminants are present. Proper preventive measures, such as improving the integrity of the silver layer and controlling environmental conditions, are essential to mitigate these issues.
How to Mitigate the Galvani Effect in Silver-Plated PCBs?
The Galvani Effect in silver-plated PCBs can be mitigated by optimizing design, controlling manufacturing processes, applying protective coatings, and selecting appropriate materials to reduce moisture exposure and corrosion risks.
Design Optimization Solutions
To reduce the Galvani Effect in silver-plated PCBs, proper design choices are crucial.
Avoid Using Silver-Plated PCBs in High Temperature and Humidity Environments:
The Galvani Effect is more likely to occur in environments where moisture and high temperatures are present. High humidity accelerates the corrosion process by facilitating the electrochemical reaction between copper and silver. By avoiding the use of silver-plated PCBs in such conditions, designers can minimize the risk of corrosion.
Optimize the Design of Pads and Circuit Connections:
It is essential to improve the design of the pads and the connections between circuits. Design modifications, such as increasing the thickness of the silver layer or ensuring better sealing around the pads, can reduce the exposure of copper to the elements. Additionally, making sure that there are no gaps between the silver layer and the copper pad will limit the chances of electrolytes reaching the copper surface.
Process Control Measures
Manufacturing processes play a key role in controlling the Galvani Effect.
Increase Silver Layer Density and Reduce Pores:
A dense, uniform silver layer is less likely to have microscopic pores or gaps that allow moisture or electrolytes to reach the copper beneath. By optimizing the silver plating process to ensure a smoother, more compact silver layer, the risk of corrosion is reduced.
Strict Control of Cleanliness and Prevention of Contaminants:
Contaminants such as chloride or sulfur can accelerate corrosion. Ensuring that the PCB is clean and free from these harmful substances during the production process is vital. This can be achieved by using high-quality cleaning agents and techniques to remove oils, salts, and other contaminants from the PCB surface.
Optimize Solder Mask Alignment and Adhesion:
The solder mask plays a protective role in PCB manufacturing, but poor alignment or weak adhesion can create gaps or channels for moisture to reach the underlying copper. Optimizing solder mask application will reduce the likelihood of corrosion at these vulnerable points.
Post-Manufacturing Protective Measures
After the PCB is produced, additional protective steps can further reduce the risk of the Galvani Effect.
Apply Conformal Coatings to Seal Out Moisture and Contaminants:
Conformal coatings, such as protective coatings or three-proof paint (three-proofing: moisture, dust, and corrosion resistance), can create a barrier between the PCB and harmful environmental factors. By sealing the surface, these coatings prevent moisture and contaminants from reaching the copper and silver layers.
Control Storage Conditions to Minimize Environmental Impact:
Storing PCBs in dry, low-humidity environments is crucial. Moisture can accelerate the corrosion process, so controlling the temperature and humidity in storage areas can help prevent the Galvani Effect. It is important to store PCBs in sealed, moisture-resistant packaging during transportation and storage.
Key Factors in Material Selection
Selecting the right materials and ensuring compatibility with the operating environment are important steps in mitigating the Galvani Effect.
Silver Layer Thickness and Uniformity in Corrosion Resistance:
A thicker and more uniform silver layer can better resist corrosion because it provides a more substantial barrier between copper and the external environment. Inconsistent silver plating increases the risk of corrosion, as thinner or imperfect areas are more vulnerable.
Copper Substrate Treatment and Corrosion Resistance:
Copper substrates should be treated with corrosion-resistant coatings or processes to enhance their durability. Proper treatment helps to reduce the likelihood of copper oxidation, which can further exacerbate the Galvani Effect.
Compatibility with Environmental Conditions:
PCBs used in high humidity or high-temperature environments must be designed with materials that are compatible with those conditions. For example, selecting corrosion-resistant materials for both copper and silver, as well as ensuring adequate protection against environmental stressors, can prolong the lifespan of the PCB.

Conclusion
Understanding and mitigating the Galvani Effect is crucial for ensuring the longevity and reliability of silver-plated PCBs. By focusing on design optimization, process control, protective measures, and material selection, the risk of corrosion can be minimized, leading to more durable and high-performance electronic circuits.
For those seeking top-quality PCBs and PCBA solutions that prioritize reliability and precision, PCBMASTER stands out as a trusted supplier. With years of expertise in the field, PCBMASTER is committed to delivering exceptional products designed to meet the highest standards, ensuring your electronics perform at their best.
FAQs
1. How Does the Galvani Effect Impact the Lifespan of a Circuit Board?
The Galvani Effect significantly shortens the lifespan of a circuit board due to metal loss and electrical failure caused by corrosion. As copper is corroded by the galvanic reaction, the material deteriorates, weakening the board’s structure and potentially leading to functional failure, especially in areas where copper traces are involved.
2. Is a Thicker Silver Layer Safer to Prevent the Galvani Effect?
While a thicker silver layer may offer better protection against corrosion, it’s not a perfect solution. The thickness of the silver layer does help to delay the onset of galvanic corrosion by acting as a more robust barrier between copper and the environment. However, there are diminishing returns, as too thick a layer can introduce issues like higher costs or compromised solderability. It’s important to find an optimal balance for the application.
3. Why Do Silver-Plated PCBs Experience the Galvani Effect More in Humid Environments?
In humid environments, the presence of electrolytes, such as water with dissolved ions, accelerates the galvanic corrosion process. These electrolytes facilitate the electrochemical reactions between copper (acting as the anode) and silver (acting as the cathode), causing copper to corrode more rapidly. The moisture essentially triggers the galvanic effect, making it worse than in dry environments.
4. Can the Galvani Effect Be Completely Avoided?
It is challenging to completely avoid the Galvani Effect, but its impact can be significantly reduced. Through design improvements, careful process control, and post-manufacturing protective measures, the severity of the galvanic corrosion can be minimized. Preventive strategies, such as using corrosion-resistant materials or applying protective coatings, can significantly improve the longevity of silver-plated PCBs.
5. Does the Galvani Effect Impact the Electrical Performance of a Circuit Board?
Yes, the Galvani Effect does affect the electrical performance of a PCB. The corrosion products, such as copper salts and electrolytes, can increase contact resistance, leading to poor electrical conductivity. In severe cases, this can result in open circuits, further compromising the performance of the board and potentially leading to complete system failure.
Author Bio
Hi, I'm Carol, the Overseas Marketing Manager at PCBMASTER, where I focus on expanding international markets and researching PCB and PCBA solutions. Since 2020, I've been deeply involved in helping our company collaborate with global clients, addressing their technical and production needs in the PCB and PCBA sectors. Over these years, I've gained extensive experience and developed a deeper understanding of industry trends, challenges, and technological innovations.
Outside of work, I'm passionate about writing and enjoy sharing industry insights, market developments, and practical tips through my blog. I hope my posts can help you better understand the PCB and PCBA industries and maybe even offer some valuable takeaways. Of course, if you have any thoughts or questions, feel free to leave a comment below—I'd love to hear from you and discuss further!



